Our 2030 Impact.
Every number here represents a life — a family — a future saved by precision care.
Cardiac arrest survival
Certified first responders
“Every second of CPR precision is a second closer to a heartbeat.”
We didn’t set out to just build another piece of medical equipment. We set out to give every emergency responder the exact precision needed to bring a heartbeat back. This is not just engineering—this is a fight for life.

Where the Ileria mission began
After two early setbacks in the energy sector, the Ileria founding team pivoted toward healthcare — and so began its journey towards life-saving CPR technology innovation.
We identified a critical need
Driven by the urgency of cardiovascular disease, we identified resuscitation as a life-saving intervention with significant gaps. We developed an early concept for an automated CPR device.

Market-driven shift
Initial concepts failed to meet clinical expectations. Through direct market research and feedback, we identified other needs — leading to a strategic shift toward supporting manual CPR.
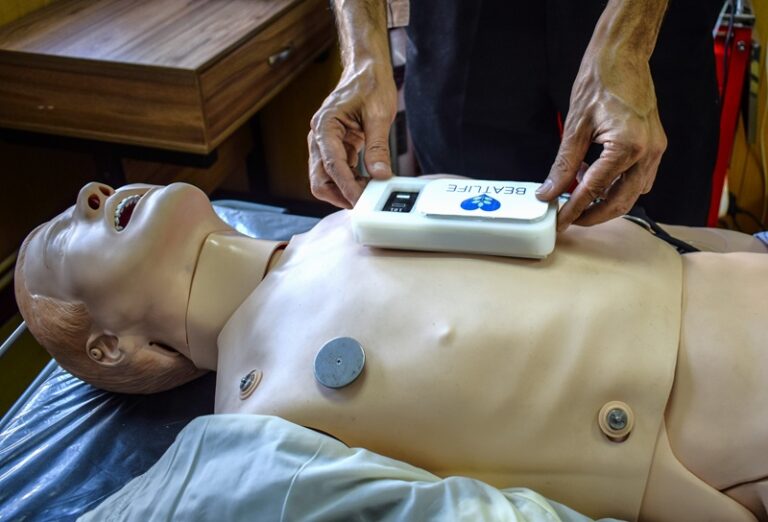
First resuscitation feedback prototype
We built our first physical prototype to support manual CPR. Designed for real-time feedback, it allowed us to test functionality and explore how best to support nurses.

Prototype v1.1
We improved the device’s physical design to enable broader market research. Version 1.1 featured a refined casing and a more intuitive structure for demonstration.
Version 2: Functional testing
We introduced the Version 2 model with a redesigned casing and a new approach to achieve better functionality. This led to Ileria’s first sales experience.

Version 3: First circuit board design
With Version 3, we designed Ileria’s first custom printed circuit board (PCB), allowing for more precise control of the device’s functions and improved reliability.

Version 4: Hardware meets software
We redesigned the device’s internal architecture due to an issue identified in V.3 — bringing significant electrical improvements and launching our first embedded OS.

Physical form confirmed
We tested over 40 printed casings to fine-tune the device’s shape, grip, and durability. These iterations helped us finalize the injection molding.

Version 5: PCB redesign
As the device’s housing changed, the PCB had to follow — and thus began a productive back-and-forth collaboration between Ileria’s engineering teams.

Version 6: Enhanced stability
We redesigned the internal PCB to reduce dependency on rare components. This introduced a new circuit layout optimized for long-term manufacturability.

Version 7: Medical device safety
During defibrillation simulations, we observed magnetic interference affecting system stability. Version 7 was designed to simulate robust safety test conditions.
Failure was not an option.
In MedTech, safety testing isn’t a formality—it is the difference between life and death. The shift to medical-grade pressure revealed vulnerabilities we had to face head-on.

Version 8: Safety under pressure
Version 8 failed a high-voltage test in simulated emergency scenarios — one of Ileria’s most frustrating days. This setback led us to discover new techniques for safety.

Version 9: Safety audit
We fine-tuned every layer. Version 9 provided stable performance over three months of continuous test scenarios, confirming its readiness for external validation.

Version 10: Ready for external testing
Developed specifically for IEC 60601-1 certification testing, it featured full electrical protection and redesigned safety systems for patient safety.

Version 10: CPR Technology Innovation success
Version 10 passed the IEC 60601-1 safety standard — one of the most significant milestones for our CPR technology innovation. We implemented complete electrical isolation.

Ready for semi-mass production
After meeting all safety requirements and integrating clinical feedback, Version 11 was completely optimized for scalable, semi-mass production lines.
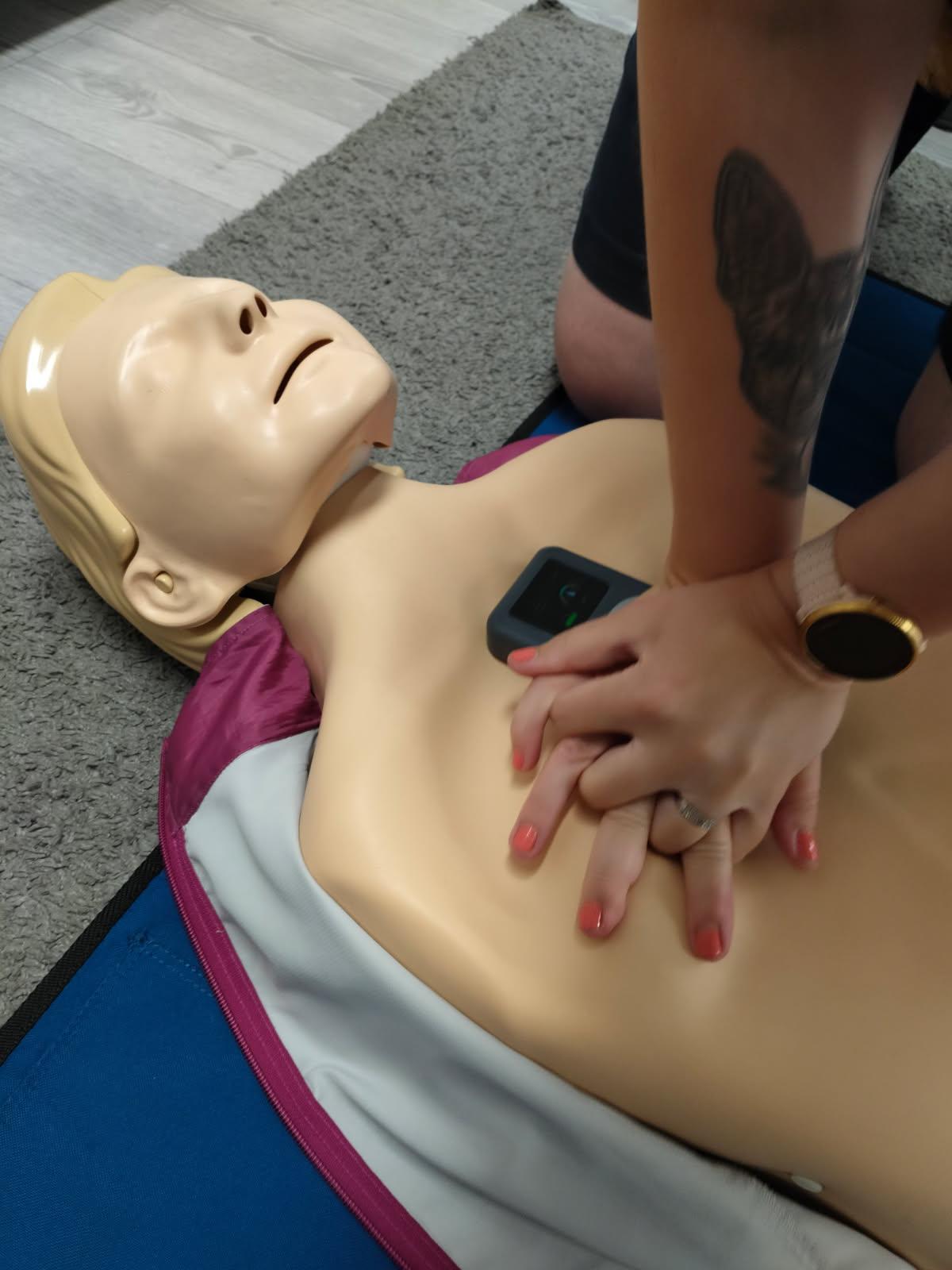
Version 11.1: User pilots
We launched the first user pilots in real-world resuscitation scenarios, proving our CPR technology innovation works under high pressure in hospital environments.

Version 11.2: Product ready
Based on pilot results, we made firmware refinements and optimized the user interface for maximum speed. The product is now ready for wide-scale deployment.

Ileria AI: Advanced CPR Technology Innovation
The Ileria ecosystem expands — Ileria AI is now live. It is an advanced, data-driven coach that analyzes resuscitation performance through cutting-edge CPR technology innovation.
The journey of CPR technology innovation continues.
After years of engineering, testing, and perfecting, the foundation is set. We are no longer just building technology—we are preparing to save 200,000 lives.
On the path to medical-grade.
IEC 60601-1
Designed to comply with international standards for basic safety and essential performance of medical electrical equipment.
ISO 13485 Ready
Applying Quality Management Systems (QMS) specifically tailored for the design and manufacture of high-reliability medical devices.
Discover the medical-grade device built directly for those experiencing cardiac arrest.
